Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (10): 1617-1625.doi: 10.3969/j.issn.2095-4344.1610
Previous Articles Next Articles
Application of bisphosphonates incorporated into scaffolds in bone defects
Cui Yutao, Li Ronghang, Liu He, Wang Zhonghan, Li Shengyang, Ji Xuan, Yang Fan, Guan Wenqi, Li Zuhao, Wu Dankai
- Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2018-11-21Online:2019-04-08Published:2021-04-28 -
Contact:Wu Dankai, Chief physician, Professor, Master’s supervisor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Cui Yutao, Master candidate, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 81671804, 81772456 and 81171681 (all to LH)
CLC Number:
Cite this article
Cui Yutao, Li Ronghang, Liu He, Wang Zhonghan, Li Shengyang, Ji Xuan, Yang Fan, Guan Wenqi, Li Zuhao, Wu Dankai . Application of bisphosphonates incorporated into scaffolds in bone defects[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(10): 1617-1625.
share this article

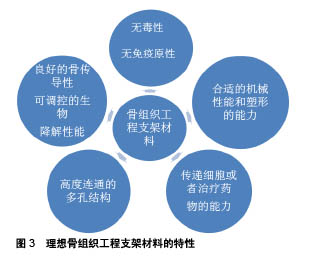
2.1 骨组织工程支架 骨组织工程是一种组织再生技术,其通过生物材料提供修复初期的机械性能,同时通过在局部持续释放生长因子、药物促进缺损部位骨的再生修复,其主要包括3个重要因素:种子细胞、支架材料和生长因子。支架材料作为骨组织工程的基本要素之一,其最理想的条件:首先要具有生物相容性,在生物体内应该是无毒性的,这是前提条件;其次,材料还应当具有卓越的骨传导性、可调控的生物降解性能、高度连通的多孔结构、传递细胞或治疗药物的能力、合适的机械性能和塑形能力等,见图3[24-25]。用于骨组织再生的当代生物材料可大致分为无机和有机材料。无机材料,如β-磷酸三钙、羟基磷灰石和生物活性玻璃等,长期以来一直用于骨组织工程,因为它们在结构和组成上与骨本身相似,无机生物材料的好处之一是它们的抗压强度和潜在的骨传导性,但他们的主要缺点是脆性。"
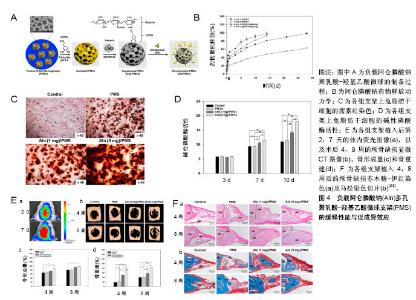
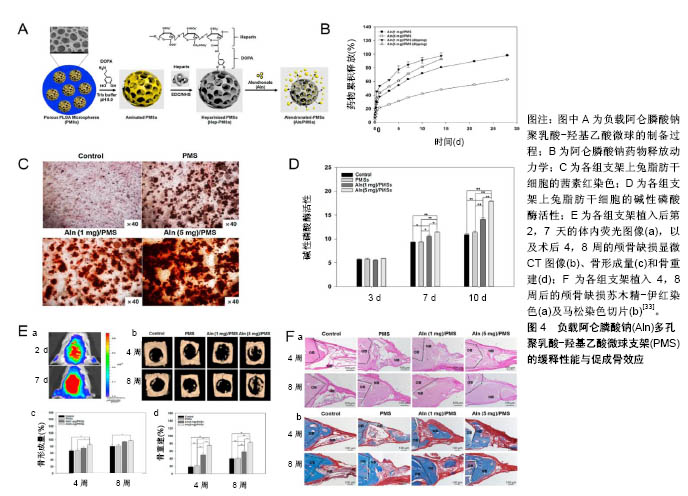
有机生物材料包括天然存在的材料和化学合成的材料,天然存在的材料包括透明质酸、壳聚糖、纤维素等,这些材料的特征在于生物相容性,使得细胞能够在其结构内黏附和迁移,主要限制在于加工和纯化方面的困难及免疫原性问题。由于天然存在材料的相关限制,有机聚合物的合成在骨组织工程显著增加,经常用于骨组织再生的聚合物包括聚乳酸、聚乙醇酸、聚乳酸羟基乙酸、聚己内酯等,他们大多拥有良好的生物相容性、生物降解性,无细胞毒性,但也有些聚合物缺乏生物活性,甚至有些降解产物包括可能阻碍再生过程的酸性副产物[26]。所以,虽然在缺损局部形成药物缓释系统逐渐成为治疗临界骨缺损的基本要求,但合适的载体材料是骨组织工程支架负载药物在骨缺损局部发挥成骨效应、促进缺损修复的必要条件[27]。 除了载体材料,载体与药物的负载方式、药物剂量及作用时间等均会对药物在缺损局部缓释及发挥成骨效应产生影响。其中药物的负载方式主要有制作混合物复合支架(如制作水凝胶支架)、药物表面涂层、药物浸泡等。药物的负载受到支架吸附能力、孔隙大小、孔隙率等较多因素的影响,这些因素会影响应用于局部的药物剂量与药物在局部的释放动力学,可能对实验结果产生较大影响。 2.2 双膦酸盐类药物复合支架 在基于细胞活性药物的骨组织工程领域,已形成了一种共识:即在生理水平上持续递送细胞活性药物的合适载体,对于其在骨修复中的成骨功效至关重要[28]。双膦酸盐类药物在骨缺损局部的应用,主要是与生物材料合成双膦酸盐类药物复合支架。通过3D打印等技术可定制复合支架的形状及结构,将体积与骨缺损部位相当的复合支架完全填充于缺损部位,这有利于在缺损局部提供良好的力学支持,并形成良好的局部药物缓释系统,使双膦酸盐类药物可在缺损局部持续浓集,达到加速新骨形成、促进缺损愈合的预期效果。这一过程中,合适的载体将是双膦酸盐类药物在缺损局部发挥成骨效应的关键因素。目前应用于双膦酸盐类药物复合支架的材料也主要分为有机材料和无机材料。 2.2.1 有机材料支架 有机材料是负载双膦酸盐类药物复合支架的主要组成部分。采用发泡技术、3D打印技术等制作多孔有机材料支架,再通过孔隙内置药或药物表面涂层、药物浸泡等方法使支架负载双膦酸盐类药物,植入缺损局部,通过药物在局部持续缓慢的释放而发挥作用。 聚乳酸-羟基乙酸:常被用作负载双膦酸盐类药物的生物材料,由于它具有无毒性、良好的生物相容性及可生物降解的特性和公认的药物递送特性,是FDA批准临床应用的安全生物材料[29-31]。聚乳酸-羟基乙酸负载药物可持续释放数周甚至数月,所以将其和双膦酸盐类药物相结合制作复合支架,用于缺损局部,以促进新骨形成及骨整合,是常用的载体药物缓释系统[32]。负载1,5 mg阿伦膦酸钠的多孔聚乳酸-羟基乙酸微球支架可在较长时间内持续释放药物,明显促进兔脂肪干细胞的成骨分化,将支架植入大鼠颅骨临界缺损模型4周后相比于未载药支架,载药支架明显促进大鼠颅骨缺损模型新骨的再生,并且负载5 mg阿伦膦酸钠载药支架的促成骨效应更优,见图4[33]。以胶原为致孔剂,将应用溶剂诱导相转化技术制作的3D多孔聚乳酸-羟基乙酸支架负载利塞膦酸盐后,可促进人骨肉瘤细胞的增殖,提升其钙沉积及碱性磷酸酶活性[34]。Wang等[35]制作了一种具有短期控释性能的负载阿伦膦酸钠的聚乳酸-羟基乙酸支架,并将其填充于大鼠股骨远端中轴线上的临界缺损,研究其对骨缺损愈合的影响,发现相比于植入普通聚乳酸-羟基乙酸,负载20-40 μg阿伦膦酸钠的聚乳酸-羟基乙酸在第6周时新骨形成量几乎是空白支架组和空白缺损组的2倍,其可明显促进新骨形成,提高生物力学性能。这表明双膦酸盐类药物用作骨缺损局部的缓释药物,可增强骨缺损局部的新骨形成,促进缺损修复,并且聚乳酸-羟基乙酸是双膦酸盐类药物的有效载体,负载双膦酸盐类药物的聚乳酸-羟基乙酸可在骨缺损局部形成药物缓释系统,发挥促进骨修复的功效。"
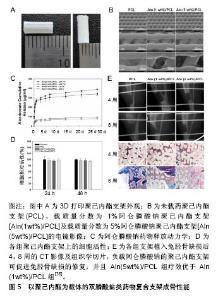
聚己内酯:具有生物降解性及良好的生物相容性,成为支架材料研究的热点。然而,由于固有的疏水性和生物活性官能团的缺乏,聚己内酯的表面特征不利于细胞黏附和增殖。有研究表明,单独应用聚己内酯支架不能显著增强兔脂肪干细胞、牙周韧带细胞和骨肉瘤细胞(MG-63)细胞的增殖和成骨分化[36],但负载成骨诱导因子可为聚己内酯在骨缺损局部应用中提供新的功能。通过3D打印制作的负载阿仑膦酸钠聚己内酯支架,可明显提升MG-63细胞的碱性磷酸酶的活性及钙含量,并在 28 d内持续有阿仑膦酸钠的释放,将其植入正常大鼠胫骨临界缺损后,第4周可明显促进缺损部位的矿化及新骨形成,并且这些效应存在阿仑膦酸钠剂量依赖性,5%阿仑膦酸钠组疗效优于1%阿仑膦酸钠组,载药组明显优于空支架组,见图5[36]。"
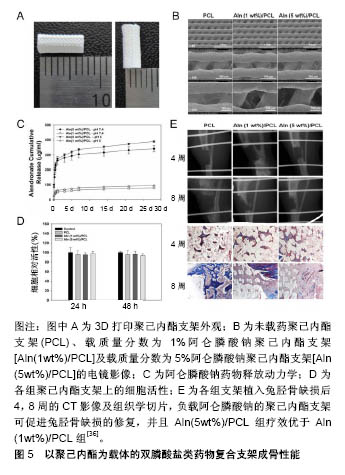
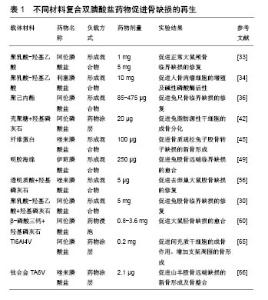
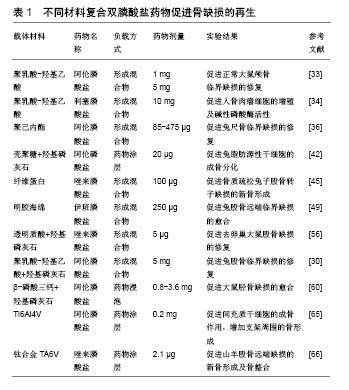
除了单纯的聚己内酯支架负载双膦酸盐类药物,研究者也经常将聚己内酯与羟基磷灰石联合制作复合材料后负载药物,使载体拥有更好的性能。Song等[37]通过静电纺丝技术制作了3D多孔聚己内酯/羟基磷灰石复合材料支架,并应用药物浸泡方法负载阿仑膦酸钠后进行细胞实验,发现其可明显促进人胎儿成骨细胞成骨相关基因的表达。然而Valente等[38]的研究则得出了不同的结果,他们对比分析了羟基磷灰石+聚己内酯+阿仑膦酸钠复合材料和不含阿仑膦酸钠羟基磷灰石+聚己内酯复合材料对兔尺骨鹰嘴临界骨缺损修复影响的组织学特征,发现两者对于缺损修复的影响并无明显差别,负载阿仑膦酸钠对羟基磷灰石+聚己内酯复合材料的成骨效应无明显影响。这些结果的差异可能与阿仑膦酸钠的作用时间、支架制作工艺等相关,但聚己内酯负载阿仑膦酸钠仍然是一种促进骨缺损局部愈合很有潜质的方法。 壳聚糖:是一种天然生物材料,是骨组织工程常用的药物递送载体,它具有良好的生物相容性、无毒性、生物降解性、抗菌活性。此外,壳聚糖是自然界中第二丰富的多糖,这使得其在生物工程和医药应用中具有很好的经济性[39-40]。但仅由壳聚糖作为载体具有明显的缺点,例如缺乏骨结合生物活性和峰值释放问题。通过将无机材料结合到复合材料中,例如生物活性玻璃、羟基磷灰石、磷酸三钙等可最小化这些缺点。其中,羟基磷灰石是骨缺损治疗最有利的材料,也是人体骨骼的主要无机成分[41],另外其可与双膦酸盐类药物分子强烈结合,保证最佳的药物负载。Wu等[42]将壳聚糖和羟基磷灰石-阿仑膦酸钠制作混合物形成复合微球体系,作为药物缓释系统进行细胞实验,结果显示复合体具有良好的药物负载率及缓释性能,是骨微环境局部缓释的理想选择,负载20 μg阿仑膦酸钠的复合支架可明显促进兔脂肪源性干细胞的增殖及成骨分化。随后他们又以壳聚糖为基质,结合L聚乳酸及10%羟基磷灰石-阿仑膦酸钠涂层制作了复合微球支架,体外细胞实验发现这种支架可明显促进脂肪源性干细胞的成骨分化,而将其与空白聚合物支架分别填充于正常兔桡骨大尺寸缺损后,观察到空白支架组在第2周有少量骨痂形成,第8周开始有明显新骨形成,而载药支架在第2周时即有明显的骨痂形成,在4-8周内缺损已基本完全愈合,具有明显促进缺损愈合的作用[40]。总之,以壳聚糖为基础制作的复合支架负载双膦酸盐类药物,可在缺损局部发挥明显的促进新骨形成效应。 纤维蛋白:是从人血浆中分离,由纤维蛋白原和凝血酶成分组成,其具有良好的生物相容性及生物降解性,同时可促进成骨细胞分化,并促进位于骨膜附近的成骨细胞形成新骨[43]。负载阿仑膦酸钠的复合纤维蛋白凝胶用于兔颅骨缺损模型局部治疗,可明显促进新骨的形成、缺损的愈合,并可促进人骨髓间充质干细胞的增殖及成骨分化[44]。Jing等[45]以纤维蛋白为材料负载唑来膦酸盐及骨形态发生蛋白用于骨质疏松兔子股骨转子缺损的治疗,结果显示负载唑来膦酸盐的纤维蛋白可明显促进缺损局部骨再生,并且唑来膦酸盐与骨形态发生蛋白联合应用时可明显增强骨形态发生蛋白在缺损局部的成骨效应。 其他材料:除了上述主要的双膦酸盐类药物复合支架有机材料外,还有研究以其他聚合物材料、天然材料及复合材料作为载体的研究。Kanoriya等[46]在纤维蛋白膜上加上血小板浓缩物,形成富血小板纤维蛋白作为载体材料,富含血小板的纤维蛋白是浓缩血小板的储存库,提供了生物生长促进因子和细胞因子的库,有助于介导骨的再生和软组织成熟,其负载双膦酸盐类药物用于缺损局部,也可产生良好的成骨效应。 天然材料透明质酸是细胞外基质的主要成分,其负载阿仑膦酸钠及骨形态发生蛋白2形成药物共释放系统,可明显提升成骨细胞成骨相关基因的表达,抑制破骨细胞介导的骨吸收[47]。阿仑膦酸钠磷酸化的Ⅰ型胶原支架,可明显促进大鼠颅骨缺损新骨的形成[48]。负载伊班膦酸钠的明胶海绵复合支架,可促进兔股骨远端缺损的修复[49]。此外,还有研究以聚丙交酯和聚乙交酯[50]、海藻酸盐等为材料作为双膦酸盐类药物的载体[51],均显示出了良好的成骨促进效应及作为双膦酸盐类药物复合支架的潜力。 总之有机材料是双膦酸盐类药物复合支架的研究热点,因为他们具有的良好的生物相容性及降解特性,其负载双膦酸盐类药物可在局部形成良好的药物缓释系统,虽然有机材料的机械性能不足等也可能会增加缺损修复初期的局部不稳定性,但多数实验结果也证实以有机材料为基础制作的双膦酸盐类药物复合支架,可在缺损局部达到预期的促进成骨、促进缺损愈合效应。因此以有机材料为基础的双膦酸盐类药物复合支架,可能是用于局部骨缺损治疗未来的临床治疗策略之一。 2.2.2 无机材料 无机材料大多与骨组织具有一定的相似性,因其良好的抗压能力、骨传导性,常被用作双膦酸盐类药物复合支架的材料。负载双膦酸盐类药物的无机材料载体,以羟基磷灰石、磷酸钙及金属支架最为广泛。 羟基磷灰石:是人体骨骼的主要无机成分,被广泛用作骨修复和骨组织再生的生物材料,其主要优势在于:具有良好的生物相容性及骨传导性,且无免疫原 性[52-53],而且羟基磷灰石的磷酸钙晶体可与阿仑膦酸钠分子强烈结合,产生最佳的阿仑膦酸钠负载。有研究显示羟基磷灰石本身也具有促进骨整合的能力,但其机械性能差及降解速度慢是主要缺点[38]。单独应用羟基磷灰石作为双膦酸盐类药物载体的研究并不多见。Boanini等[54]分别将不同浓度的阿仑膦酸钠与羟基磷灰石制作成混合纳米材料,并将人成骨细胞及MG63细胞种植于混合材料上培养,发现混合材料可促进细胞的碱性磷酸酶、Ⅰ型胶原和骨钙素的合成,并且阿仑膦酸钠含量高组的效应更加明显。但相反,Canettieri等[55]分别将1 mol和2 mol的阿仑膦酸钠负载于羟基磷灰石,制作载药羟基磷灰石复合支架,将其植入去卵巢大鼠股骨缺损后第21天发现,相比于对照组,羟基磷灰石-阿仑膦酸钠复合支架会抑制新骨的形成。不过这一结果的形成可能是因为阿仑膦酸钠的剂量过大,产生了对成骨抑制的作用。 研究者们更趋向于用羟基磷灰石和其他材料复合形成药物载体,克服羟基磷灰石的一些缺点,更好地形成局部缓释。由于有机材料常有着良好的生物降解性,包括聚乳酸-羟基乙酸、聚己内酯、壳聚糖等,常与羟基磷灰石形成复合材料作为药物载体[38]。多数研究结果也证实了羟基磷灰石与有机材料形成复合材料作为载体负载双膦酸盐类药物,可到达促进骨再生的预期结果。比如负载阿仑膦酸钠的聚乳酸-羟基乙酸/羟基磷灰石烧结微球支架,可明显促进骨髓间充质干细胞的成骨分化及其成骨相关基因的表达,将其植入小鼠背部皮下组织及兔股骨临界缺损后,发现其可明显促进局部新生骨的形成[30]。结合5 μg唑来膦酸盐与羟基磷灰石及透明质酸制作的水凝胶支架,可明显促进去卵巢大鼠股骨髁缺损的修复,抑制大鼠破骨细胞前体细胞的增殖[56]。另外也有研究者将羟基磷灰石与其他无机材料联合形成复合材料,但也未能达到理想的效果。负载阿仑膦酸钠的羟基磷灰石-磷酸三钙复合生物材料,联合牵张成骨治疗兔左侧胫骨临界缺损模型后,发现其对成骨效应产生抑制作用,抑制新骨形成[57]。 羟基磷灰石有良好的生物相容性、骨传导性及与双膦酸盐类药物强烈结合的性质,从目前的研究看,羟基磷灰石与有机材料复合后负载双膦酸盐类药物,能更好地达到药物促进成骨作用。 磷酸钙:具有良好的生物活性、骨组织的相似性及良好的生物降解性,常被用作骨科移植物的材 料[58]。β-磷酸三钙是磷酸钙材料中应用最为广泛的之一,其可为骨形成提供支架,也是广泛使用的生长因子和抗生素药物递送载体。在临床研究中,β-磷酸三钙负载双膦酸盐类药物的复合支架也常有应用。将负载阿仑膦酸钠的β-磷酸三钙复合药物缓释支架用于牙周骨缺损患者,临床研究发现与单独使用β-磷酸三钙相比,β-磷酸三钙负载阿仑膦酸钠的复合支架在促进骨形成方面是有效的[59]。但β-磷酸三钙支架植入体内后,其生物溶解性与破骨细胞有着很密切的关系,因此β-磷酸三钙负载双膦酸盐类药物后,双膦酸盐类药物对破骨性骨溶解的抑制作用可能会对β-磷酸三钙的生物降解也产生一定的抑制作用。Tanaka等[15]用阿仑膦酸钠浸泡β-磷酸三钙支架后,植入兔股骨髁缺损模型,结果显示阿仑膦酸钠浸泡的β-磷酸三钙支架减少了支架表面的破骨细胞数量,抑制了破骨重吸收,同时也抑制了β-磷酸三钙支架的生物溶解,甚至也导致了与剂量相关的成骨抑制。这可能是由于双膦酸盐类药物抑制了由细胞介导的β-磷酸三钙支架生物降解,从而对其成骨效应产生了影响。关于双膦酸盐类药物对β-磷酸三钙支架生物降解性的具体影响还有待进一步的探讨研究。由于单纯的磷酸钙支架存在一定的不足,有研究者将2种磷酸钙结合形成复合物,使载体拥有更好的性能。双相磷酸钙是β-磷酸三钙与羟基磷灰石的混合物,有着良好的生物降解性及生物相容性,因而也是磷酸钙材料中常用的药物载体。负载阿仑膦酸钠的双相磷酸钙支架,可在28 d内持续释放阿仑膦酸钠,提升MG-63细胞的碱性磷酸酶活性、钙沉积及成骨相关基因表达,将其植入正常大鼠胫骨内侧临界缺损模型第4,8周时,发现相比于空白支架,载药双相磷酸钙支架填充的胫骨缺损局部新骨形成量及骨密度更高[60]。 金属支架:金属材料常因其较好的强度、耐腐蚀性及骨传导性而被广泛用作骨科移植物材料[61],常用的金属材料包括钛、镁、钽等。钛及其合金是骨科移植物中应用最为广泛的金属材料,常用于各种骨折骨缺损的治疗,其具有较合适的机械强度、良好的生物相容性。但单纯钛及其合金制作的支架,有时不能与骨组织之间形成牢固的骨整合连接,容易产生支架松动,所以涂覆生物活性分子涂层做表面修饰,是促进金属植入后愈合的一种有效方法[62-64]。涂覆阿仑膦酸钠涂层的Ti6Al4V支架,可促进间充质干细胞的成骨分化及成骨相关基因的表达,增加支架周围的骨形成[65]。具有羟基磷灰石涂层的钛合金TA6V负载2.1 μg唑来膦酸盐形成的复合金属支架,可明显促进去卵巢山羊股骨远端骨骺处缺损的新骨新成及骨整合,增强钛合金支架植入后的稳定性[66]。Park等[67]将阿仑膦酸钠与万古霉素以点状分布的形式涂覆于钛接骨板上,研究发现这种接骨板表现出了明显的抗菌活性,并可促进成骨细胞的增殖。所以钛支架点状涂覆阿仑膦酸钠及万古霉素涂层,是促进骨缺损的骨形成及抗菌作用的有前途治疗方法。 多孔钽具有高摩擦系数、高孔隙率、较低的弹性模量及良好的组织相容性等特性,因而可用于植入骨缺损的材料,但同时其也具有成本较高及加工制作难度较高的相对不足,这在一定程度上限制了其在临床发面的应用发展[68]。Garbuz等[69]制作了阿仑膦酸钠-磷酸钙涂层的多孔钽移植物,将其与无涂层多孔钽移植物及磷酸钙涂层多孔钽移植物分别植入兔股骨远端临界缺损模型,发现添加了阿仑膦酸钠释放涂层的多孔钽移植物,可明显增加移植物与骨之间的缺损填充,以及新生骨的长入。镁及其合金是一种很有研究价值的可降解金属材料,其具有良好的机械性能及生物安全性,但其降解速率与骨缺损愈合及新骨形成不匹配是重要的缺陷,研究者常通过形成合金、表面修饰等方式来弥补其缺陷。Wei等[70]将锶取代的羟基磷灰石纳米晶和阿仑膦酸钠共同固定在AZ31镁合金表面,开发了一种新的治疗涂层策略,这一涂层明显提高了镁合金的耐腐蚀性,并明显促进了成骨细胞的增殖及分化。这些研究均证明了金属支架涂覆双膦酸盐类药物涂层应用于骨缺损局部治疗,可达到促进成骨的作用,是一种很有前途的治疗方法。 不同材料负载双膦酸盐类药物用于骨缺损局部的研究,见表1。"
| [1] Cottrell JA, Vales FM, Schachter D, et al.Osteogenic activity of locally applied small molecule drugs in a rat femur defect model.J Biomed Biotechnol.2010;2010:597641.[2] Conners CM, Bhethanabotla VR, Gupta VK. Concentration- dependent effects of alendronate and pamidronate functionalized gold nanoparticles on osteoclast and osteoblast viability. J Biomed Mater Res B. 2017;105(1):21-29.[3] Dimitriou R, Jones E, McGonagle D, et al. Bone regeneration: current concepts and future directions. BMC Med.2011;9:66.[4] Anglen KSGKMDTOMJO.Evolution of Bone Grafting: Bone Grafts and Tissue Engineering Strategies for Vascularized Bone Regeneration.Clinic Rev Bone Miner Metab.2015;13:232-244.[5] Zhu JY, Huang BL, Ding S,et al.Tethering of rhBMP-2 upon calcium phosphate cement via alendronate/heparin for localized, sustained and enhanced osteoactivity. Rsc Adv. 2017;7(33): 20281-20292.[6] Martin V, Bettencourt A. one regeneration: Biomaterials as local delivery systems with improved osteoinductive properties.Mat Sci Eng C-Mater.2018;82:363-371.[7] Liu Y, Wu G, de Groot K. Biomimetic coatings for bone tissue engineering of critical-sized defects. J R Soc Interface.2010;7 Suppl 5:S631-647.[8] Rachner TD,Khosla S,Hofbauer LC.Osteoporosis: now and the future. Lancet.2011;377(9773):1276-1287.[9] Hemmeler C, Morell S, Amsler F, et al.Screening for osteoporosis following non-vertebral fractures in patients aged 50 and older independently of gender or level of trauma energy-a Swiss trauma center approach. Arch Osteoporos.2017;12(1):38.[10] Papapoulos SE.Bisphosphonate actions: physical chemistry revisited. Bone. 2006;38(5):613-616.[11] Russell RG, Watts NB, Ebetino FH, et al.Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int.2008;19(6):733-759.[12] Tarafder S, Bose S. Polycaprolactone-Coated 3D Printed Tricalcium Phosphate Scaffolds for Bone Tissue Engineering: In Vitro Alendronate Release Behavior and Local Delivery Effect on In Vivo Osteogenesis. ACS Appl Mater Interfaces.2014;6(13): 9955-9965.[13] Ossipov DA. Bisphosphonate-modified biomaterials for drug delivery and bone tissue engineering. Expert Opin Drug Deliv. 2015;12(9): 1443-1458.[14] Cui P, Liu H, Sun J, et al.Zoledronate promotes bone formation by blocking osteocyte-osteoblast communication during bone defect healing. Histol Histopathol.2018;33(1):89-99.[15] Tanaka T, Saito M, Chazono M, et al.Effects of alendronate on bone formation and osteoclastic resorption after implantation of beta-tricalcium phosphate. J Biomed Mater Res A. 2010;93(2): 469-474.[16] Dutra BC, Oliveira A, Oliveira PAD, et al.Effect of 1% sodium alendronate in the non-surgical treatment of periodontal intraosseous defects: a 6-month clinical trial. J Appl Oral Sci. 2017;25(3):310-317.[17] Hu X, Neoh KG, Shi Z, et al.An in vitro assessment of fibroblast and osteoblast response to alendronate-modified titanium and the potential for decreasing fibrous encapsulation. Tissue Eng Part A. 2013; 19(17-18):1919-1930.[18] Hong C, Quach A, Lin L, et al.Local vs. systemic administration of bisphosphonates in rat cleft bone graft: A comparative study.PloS One.2018;13(1):e0190901.[19] Toker H, Ozdemir H, Ozer H, et al.A comparative evaluation of the systemic and local alendronate treatment in synthetic bone graft: a histologic and histomorphometric study in a rat calvarial defect model Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(5): S146-S52.[20] Patel V, Graves L, Lukert B.Pelvic fractures associated with long-term bisphosphonate therapy - case report. J Musculoskelet Neuronal Interact.2013;13(2):251-254.[21] Franceschetti P, Bondanelli M, Caruso G, et al.Risk factors for development of atypical femoral fractures in patients on long-term oral bisphosphonate therapy.Bone.2013;56(2):426-431.[22] Whyte MP, McAlister WH, Novack DV, et al. Bisphosphonate- induced osteopetrosis: Novel bone modeling defects, metaphyseal osteopenia, and osteosclerosis fractures after drug exposure ceases. J Bone Miner Res. 2008;23(10): 1698-107.[23] Toker H, Ozdemir H, Ozer H, et al. Alendronate enhances osseous healing in a rat calvarial defect model.Arch Oral Biol. 2012;57(11): 1545-1550.[24] 金灿,陈振琦.应用3D打印技术制作组织工程支架:修复骨缺损的研究回顾[J].中国组织工程研究,2017,21(10):1611-1616.[25] Li YH, Wang ZD, Wang W,et al.The biocompatibility of calcium phosphate cements containing alendronate-loaded PLGA microparticles invitro.Exp Biol Med.2015;240(11):1465-1471.[26] Shrivats AR, McDermott MC, Hollinger JO.Bone tissue engineering: state of the union. Drug Discov Today. 2014;19(6): 781-786.[27] Chen J, Luo Y, Hong L, et al.Synthesis, characterization and osteoconductivity properties of bone fillers based on alendronate-loaded poly(epsilon-caprolactone)/hydroxyapatite microspheres. J Mater Sci Mater Med.2011;22(3):547-555.[28] Cartmell S. Controlled release scaffolds for bone tissue engineering. J Pharm Sci.2009;98(2):430-441.[29] Wang CZ, Chen SM, Chen CH, et al.The effect of the local delivery of alendronate on human adipose-derived stem cell-based bone regeneration. Biomaterials. 2010;31(33):8674-8683.[30] Shi XT, Ren L, Tian M, et al.In vivo and in vitro osteogenesis of stem cells induced by controlled release of drugs from microspherical scaffolds. J Mater Chem.2010;20(41):9140-9148.[31] Posadowska U, Parizek M, Filova E, et al.Injectable nanoparticle-loaded hydrogel system for local delivery of sodium alendronate.Int J Pharmaceut.2015;485(1-2):31-40.[32] Limirio PHJO, Rocha FS, Batista JD, et al.The Effect of Local Delivery Doxycycline and Alendronate on Bone Repair. AAPS Pharm Sci Tech. 2016;17(4):872-877.[33] Lee JY, Kim SE, Yun YP, et al. Osteogenesis and new bone formation of alendronate-immobilized porous PLGA microspheres in a rat calvarial defect model.J Ind Eng Chem.2017;52:277-286.[34] Elkasabgy NA, Mahmoud AA, Shamma RN.Determination of cytocompatibility and osteogenesis properties of in situ forming collagen-based scaffolds loaded with bone synthesizing drug for bone tissue engineering. Int J Polym Mater Po. 2018;67(8): 494-500.[35] Wang YH, Rajalakshmanan E, Wang CK, et al. PLGA-linked alendronate enhances bone repair in diaphysis defect model. J Tissue Eng Regen M. 2017;11(9):2603-2612.[36] Kim SE, Yun YP, Shim KS, et al.3D printed alendronate-releasing poly(caprolactone) porous scaffolds enhance osteogenic differentiation and bone formation in rat tibial defects.Biomed Mater. 2016;11(5):055005.[37] Song JQ, Zhu GL, Wang L,et al.Assembling of electrospun meshes into three-dimensional porous scaffolds for bone repair. Biofabrication. 2017;9(1):015018.[38] Valente FL,Reis ECC,Sepulveda RV,et al.Hydroxyapatite, polycaprolactone and alendronate composites for bone regeneration in rabbits' olecranon: histological features. Arq Bras Med Vet Zoo. 2016;68(2):543-547.[39] Dash M, Chiellini F, Ottenbrite RM, et al.Chitosan-A versatile semi-synthetic polymer in biomedical applications. Prog Polym Sci. 2011;36(8):981-1014.[40] Wu HW, Lei PF, Liu GY, et al.Reconstruction of Large-scale Defects with a Novel Hybrid Scaffold Made from Poly(L-lactic acid)/Nanohydroxyapatite/Alendronate-loaded Chitosan Microsphere: in vitro and in vivo Studies. Sci Rep.2017;7(1):359.[41] Ding CC, Teng SH, Pan H.In-situ generation of chitosan/ hydroxyapatite composite microspheres for biomedical application. Mater Lett.2012;79:72-74.[42] Wu H, Xu Y, Liu G,et al.Emulsion cross-linked chitosan/ nanohydroxyapatite microspheres for controlled release of alendronate. J Mater Sci Mater Med. 2014;25(12):2649-2658.[43] Janmey PA, Winer JP, Weisel JW. Fibrin gels and their clinical and bioengineering applications. J R Soc Interface. 2009;6(30):1-10.[44] Kim BS, Shkembi F, Lee J.In Vitro and In Vivo Evaluation of Commercially Available Fibrin Gel as a Carrier of Alendronate for Bone Tissue Engineering. Biomed Res Int. 2017;2017:6434169.[45] Jing D, Hao XG, Xu F, et al.Effects of local delivery of BMP2, zoledronate and their combination on bone microarchitecture, biomechanics and bone turnover in osteoporotic rabbits.Sci Rep. 2016;6:28537. [46] Kanoriya D, Pradeep AR, Garg V,et al.Mandibular Degree II Furcation Defects Treatment With Platelet-Rich Fibrin and 1% Alendronate Gel Combination: A Randomized Controlled Clinical Trial.J Periodontol.2017;88(3):250-258.[47] Zhang YR, Hu Y, Luo Z,et al.Simultaneous delivery of BMP-2 factor and anti-osteoporotic drugs using hyaluronan-assembled nanocomposite for synergistic regulation on the behaviors of osteoblasts and osteoclasts in vitro. J Biomater Sci Polym Ed. 2015;26(5):290-310.[48] He YC, Zhu T, Liu L,et al.Modifying collagen with alendronate sodium for bone regeneration applications.Rsc Adv. 2018;8(30): 16762-16772.[49] Yang ZY, Chen W, Xia ZD, et al.Local Application of Ibandronate/ Gelatin Sponge Improves Osteotomy Healing in Rabbits. PloS One. 2015;10(5):e0125807.[50] Hur W, Park M, Lee JY, et al. Bioabsorbable bone plates enabled with local, sustained delivery of alendronate for bone regeneration. J Control Release.2016;222:97-106.[51] Cattalini JP,Roether J,Hoppe A,et al.Nanocomposite scaffolds with tunable mechanical and degradation capabilities: co-delivery of bioactive agents for bone tissue engineering. Biomed Mater. 2016;11(6):065003.[52] Huang J, Lin YW, Fu XW, et al.Development of nano-sized hydroxyapatite reinforced composites for tissue engineering scaffolds. J Mater Sci Mater Med.2007;18(11):2151-2157.[53] Yang H, Gao H, Wang Y.Hollow hydroxyapatite microsphere: a promising carrier for bone tissue engineering. J Microencapsul. 2016:1-6.[54] Boanini E, Torricelli P, Gazzano M, et al. Alendronate- hydroxyapatite nanocomposites and their interaction with osteoclasts and osteoblast-like cells. Biomaterials. 2008;29(7): 790-796.[55] Canettieri ACV, Colombo CED, Chin CM, et al.Femur bone repair in ovariectomized rats under the local action of alendronate, hydroxyapatite and the association of alendronate and hydroxyapatite. Int J Exp Pathol. 2009;90(5):520-526.[56] Kettenberger U,Luginbuehl V, Procter P,et al.In vitro and in vivo investigation of bisphosphonate-loaded hydroxyapatite particles for peri-implant bone augmentation. J Tissue Eng Regen M. 2017; 11(7): 1974-1985.[57] Ni M, Li G, Tang PF, et al. rhBMP-2 not alendronate combined with HA-TCP biomaterial and distraction osteogenesis enhance bone formation. Arch Orthop Trauma Surg. 2011;131(11):1469-1476.[58] Tarafder S, Bose S.Polycaprolactone-coated 3D printed tricalcium phosphate scaffolds for bone tissue engineering: in vitro alendronate release behavior and local delivery effect on in vivo osteogenesis. ACS Appl Mater Interfaces. 2014;6(13):9955-9965.[59] Naineni R, Ravi V, Subbaraya DK, et al.Effect of Alendronate with beta - TCP Bone Substitute in Surgical Therapy of Periodontal Intra-Osseous Defects: A Randomized Controlled Clinical Trial. J Clin Diagn Res.2016;10(8):ZC113-117.[60] Park KW, Yun YP, Kim SE, et al.The Effect of Alendronate Loaded Biphasic Calcium Phosphate Scaffolds on Bone Regeneration in a Rat Tibial Defect Model.Int J Mol Sci. 2015;16(11):26738-26753.[61] Villaca JC, da Silva LCRP, de Alexandria AK, et al.Development and characterization of clay-polymer nanocomposite membranes containing sodium alendronate with osteogenic activity.Appl Clay Sci. 2017;146:475-486.[62] Kim SE, Yun YP, Park K,et al.The effects of functionalized titanium with alendronate and bone morphogenic protein-2 for improving osteoblast activity. Tissue Eng Regen Med.2013;10(6):353-361.[63] Kapat K, Srivas PK, Rameshbabu AP, et al.Influence of Porosity and Pore-Size Distribution in Ti6Al4 V Foam on Physicomechanical Properties, Osteogenesis, and Quantitative Validation of Bone Ingrowth by Micro-Computed Tomography. ACS Appl Mater Interfaces. 2017;9(45):39235-39248.[64] Bosco R, Iafisco M, Tampieri A, et al.Hydroxyapatite nanocrystals functionalized with alendronate as bioactive components for bone implant coatings to decrease osteoclastic activity.Appl Surf Sci. 2015; 328:516-524.[65] Rojo L, Gharibi B, McLister R,et al.Self-assembled monolayers of alendronate on Ti6Al4V alloy surfaces enhance osteogenesis in mesenchymal stem cells.Sci Rep. 2016;6:30548.[66] Stadelmann VA, Gauthier O, Terrier A, et al.Implants Delivering Bisphosphonate Locally Increase Periprosthetic Bone Density in an Osteoporotic Sheep Model. A Pilot Study.Eur Cells Mater. 2008;16: 10-16.[67] Park CG, Park M, Kim BH, et al.Pattern-Coated Titanium Bone Fixation Plate for Dual Delivery of Vancomycin and Alendronate. Macromol Res. 2017;25(7):756-762.[68] Levine B, Della Valle CJ, Jacobs JJ.Applications of porous tantalum in total hip arthroplasty. J Am Acad Orthop Surg. 2006; 14(12):646-655.[69] Garbuz DS, Hu Y, Kim WY,et al.Enhanced gap filling and osteoconduction associated with alendronate-calcium phosphate- coated porous tantalum. J Bone Joint Surg Am. 2008;90(5): 1090-1100.[70] Wei PB, Wang B, Lu X, et al.Bio-inspired immobilization of strontium substituted hydroxyapatite nanocrystals and alendronate on the surface of AZ31 magnesium alloy for osteoporotic fracture repair. Surf Coat Tech. 2017;313:381-390. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Zeng Xianghong, Liang Bowei. A new strategy for the treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 431-437. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||